Advertisements
Advertisements
Questions
What is a polyamide?
Briefly describe the following term giving one example:
Polyesters
Advertisements
Solution 1
The polymers having amide linkages are called polyamides. They are produced through condensation polymerization of aminocarboxylic acids or dibasic acids with diamines. Nylons are a frequent term for polyamides.
Solution 2
The polymers having amide linkages are called polyamides. They are produced through condensation polymerization of aminocarboxylic acids or dibasic acids with diamines. Nylons are a frequent term for polyamides.
The total amount of carbon atoms found in the components that make up a repeating unit is how nylons are termed. For instance, nylon-66, often known as nylon six, six, is made from the monomers hexamethylenediamine and adipic acid, each of which has six C atoms. Similarly, nylon-610 (also known as nylon six, ten) is made from hexamethylenediamine and sebacic acid, which have six and ten carbon atoms, respectively. Below is a description of several significant nylons.
Nylon-66: It is polyhexamethyleneadipamide and is a polymer of hexamethylenediamine and adipic acid.
It is prepared by condensation polymerization of hexamethylenediamine with adipic acid. The amount of time and temperature of the procedure determine the chain length.
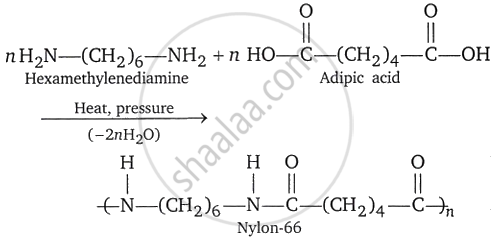
In reality, when a polysalt is produced, haxamethylenediamine and adipic acid are combined in an equimolar ratio. In an autoclave, the polysalt is dissolved in water and heated to 493K while under steam pressure. Finally, to produce nylon-66, it is heated to 543-553K. After that, it goes through a spinneret to be cast into sheets or fibers.
Nylon-610: It is a polymer of haxamethylenediamine and sebacic acid. It is prepared by the condensation polymerisation of hexamethylenediamine and sebacic acid.
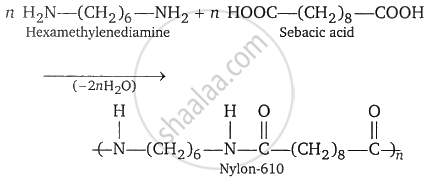
The properties and uses of nylon-610 are similar to those of nylon-66.
Nylon-6: It is also known as Perlon.
It is made by polymerizing the monomer caprolactum, which is derived from the petrochemical cyclohexane. This is how the procedure is carried out.
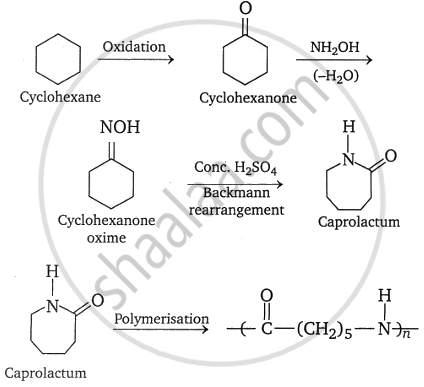
Nylon-6 filaments are produced by melt-spinning the polymer. A jet of air cools the fibers after molten polymer is pushed through a spinneret.
Notes
Students should refer to the answer according to their questions.
