Advertisements
Advertisements
Question
What happens when thin copper leaves are thrown in jar containing chlorine?
Advertisements
Solution
When thin copper leaves are thrown in a jar of chlorine they catch fire and form cupric
chloride
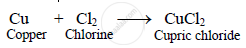
APPEARS IN
RELATED QUESTIONS
Give the reason for bleaching action of Cl2.
Write balanced equations for chlorine gas is passed into a solution of NaI in water.
Arrange the following oxyacids of chlorine – HClO, HClO2, HClO3, and HClO4 with respect to Increasing order of thermal stability.
What is the action of chlorine (Cl) on the following:
Hot and concentrated caustic soda
What is the action of chlorine (Cl) on the following:
Potassium bromide solution
Arrange the following oxyacids of chlorine - HClO, HClO2, HClO3, and HClO4 with respect to increasing order of oxidizing power.
Write any two uses of Chlorine.
What is the action of chlorine on CS2
What is the action of hydrochloric acid on NH3?
What is the action of hydrochloric acid on Na2CO3?
Answer the following.
Give two uses of HCl.
The gas evolved when sulfuric acid reacts with copper metal _______.
When SO2 is passed through an aqueous solution of I2, the solution becomes _______.
What is the action of chlorine on hot and concentrated sodium hydroxide?
What happens when chlorine reacts with?
Al
What happens when chlorine reacts with?
Na
What happens when chlorine reacts with?
P4
H2S does not produce metallic sulphide with:
Chlorine acts as a bleaching agent only in presence of ____________.
Assertion: \[\ce{NaCl}\] reacts with concentrated \[\ce{H2SO4}\] to give colourless fumes with pungent smell. But on adding \[\ce{MnO2}\] the fumes become greenish yellow.
Reason: \[\ce{MnO2}\] oxidises \[\ce{HCl}\] to chlorine gas which is greenish yellow.
When chlorine water is exposed to sunlight, the colour change which occurs is form:-
When chlorine is passed over dry slaked lime at room temperature, the product formed is ______
Complete the following reaction:
\[\ce{Na2SO3 + Cl2 + H2O -> \underline{}\underline{}\underline{}\underline{} + \underline{}\underline{}\underline{}\underline{}}\]
What is the action of hydrochloric acid on the following ?
\[\ce{Na2CO3}\]
Write the chemical reaction when chlorine reacts with dry slaked lime.
What is the action of hydrochloric acid on the NH3?
What is the action of hydrochloric acid on the following NH3?
What is the action of hydrochloric acid on Na2CO3?
What is the action of hydrochloric acid on the \[\ce{NH3}\]?
What is the action of hydrochloric acid on \[\ce{Na2CO3}\]?
What is the action of hydrochloric acid on NH3?
What is the action of hydrochloric acid on Na2CO3?
What is the action of hydrochloric acid on NH3?
What is the action of hydrochloric acid on Na2CO3?
What is the action of hydrochloric acid on \[\ce{NH3}\]?
What is the action of hydrochloric acid on \[\ce{Na2CO3}\]?
What is the action of hydrochloric acid on \[\ce{Na2CO3}\]?
What is the action of hydrochloric acid on the following?
\[\ce{Na2CO3}\]
