Advertisements
Advertisements
Question
What happens when a concentrated solution of sodium chloride (brine) is electrolysed? Write the equation of the reaction involved.
Advertisements
Solution
When electricity is passed through a concentrated solution of sodium chloride called brine, the solution decomposes to form sodium hydroxide, chlorine and hydrogen.
`2 NaCl(aq) + 2 H_2O (l)`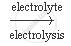 `2 NaOH (aq) + Cl_2 (g) + H_2 (g)`
`2 NaOH (aq) + Cl_2 (g) + H_2 (g)`
shaalaa.com
Is there an error in this question or solution?
