Advertisements
Advertisements
Question
What are hydrocarbons? Write the general formula of (i) saturated hydrocarbons, and (ii) unsaturated hydrocarbons and draw the structure of one hydrocarbon of each type. How can an unsaturated hydrocarbon be made saturated?
Advertisements
Solution
The compounds entirely consisting of carbons and hydrogens are known as Hydrocarbons. There are different categories in which hydrocarbons are divided out of which the two are:
(i) Saturated Hydrocarbons: The compounds of carbon having only single bonds between the carbon atoms are called saturated compounds. This includes alkanes, having a general formula CnH2n+2.
(ii) Unsaturated Hydrocarbons: The compounds of carbon having double and triple bonds between the carbon atoms are called unsaturated compounds. This includes alkenes and alkynes having general formula CnH2n and CnH2n-2, respectively.
Example
(i) Saturated hydrocarbon: C2H6 (Ethane)
Structure:
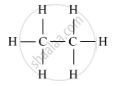
(i) Saturated Hydrocarbons: The compounds of carbon having only single bonds between the carbon atoms are called saturated compounds. This includes alkanes, having a general formula CnH2n+2.
(ii) Unsaturated Hydrocarbons: The compounds of carbon having double and triple bonds between the carbon atoms are called unsaturated compounds. This includes alkenes and alkynes having general formula CnH2n and CnH2n-2, respectively.
Example
(i) Saturated hydrocarbon: C2H6 (Ethane)
Structure:

(ii) (a) Unsaturated hydrocarbon: C2H4 (Ethene)
Structure:
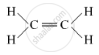
Structure:

(b) Unsaturated hydrocarbon: C2H2 (Ethyne)
Structure:
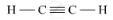
Structure:
shaalaa.com
Is there an error in this question or solution?
