Advertisements
Advertisements
Question
What is the action of bromine in alkaline medium on
i. CH3CH2NO2
ii. 
Advertisements
Solution
(a)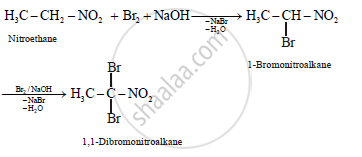
(b)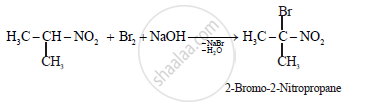
APPEARS IN
RELATED QUESTIONS
The preparation of alkyl fluoride from alkyl chloride, in presence of metallic fluorides is known as ______________.
Draw the structures of major monohalo products in each of the following reactions :

Draw the structure of major monohalo product in each of the following reactions :
The following are the possible structural isomers expected to be formed if one of the hydrogen atoms is replaced by chlorination. The original compound is
i. (CH3)2CHCH2CH2Cl
ii. (CH3)2CHCH(Cl)CH3
iii. (CH3)2C(Cl)CH2CH3
iv. CH3CHCH2(Cl)CH2CH3
Finkelstein reaction is ______.
In the following reaction, the compound used in the reaction for synthesizing ethyl fluoride is:
______ \[\ce{+ AgF -> H3C - F + AgBr}\]
Conant Finkelstein reaction for the preparation of alkyl iodide is based upon the fact that:
What is ‘A’ in the following reaction?

Which of the following are secondary bromides?
(i) \[\ce{(CH3)2 CHBr}\]
(ii) \[\ce{(CH3)3C CH2Br}\]
(iii) \[\ce{CH3CH(Br)CH2CH3}\]
(iv) \[\ce{(CH3)2 CBrCH2CH3}\]
Alkyl fluorides are synthesised by heating an alkyl chloride/bromide in presence of ______ or ______.
(i) CaF2
(ii) CoF2
(ii) Hg2F2
(iv) NaF
Discuss the role of Lewis acids in the preparation of aryl bromides and chlorides in the dark.
Why is the solubility of haloalkanes in water very low?
tert-Butylbromide reacts with aq. \[\ce{NaOH}\] by SN1 mechanism while n-butylbromide reacts by SN2 mechanism. Why?
Some alkylhalides undergo substitution whereas some undergo elimination reaction on treatment with bases. Discuss the structural features of alkyl halides with the help of examples which are responsible for this difference.
The most stable free radical among the following is
Butene-1 may be converted to butane by reaction with
Benzyl chloride (16H5CH2Cl) can be prepared from toluene by chlorination with
The alkyl halide which does not give white precipitate with alcoholic AgNO3 solution is :-
The product of reaction of alcoholic silver nitrite with ethyl bromide are:
SN1 and SN2 product are same with
\[\begin{array}{cc}
\ce{Ph - CH - CH2 - CH2 ->[Zn - Cu][Δ] Product}\\
\phantom{..}|\phantom{......}|\phantom{....................}\\
\phantom{}\ce{Br}\phantom{....}\ce{Br}\phantom{..................}
\end{array}\]
Product of the above reaction is
Name the possible alkenes which will yield 1-chloro-1-methylcyclohexane on their reaction with HCl. Write the reactions involved.
The major product of the following reaction is:
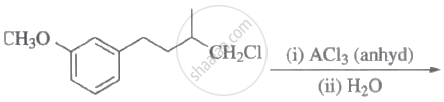
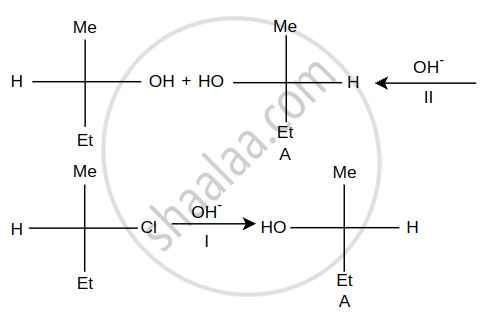
Steps I and II are ______.
