Advertisements
Advertisements
Question
Using valence bond theory, explain the hybridisation and magnetic behaviour of the following:
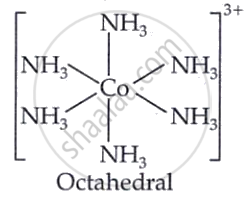
[Co(NH3)6]Cl3
(At. no.: Co = 27)
Advertisements
Solution
The complex [Co(NH3)6]Cl3 consists of a cobalt ion surrounded by six ammonia ligands, with three chloride ions as counterions. To understand its hybridisation and magnetic behaviour, we first determine the oxidation state of cobalt. Since ammonia is a neutral ligand and the overall charge on the complex ion is +3, the cobalt ion is in the +3 oxidation state, i.e., Co3+.
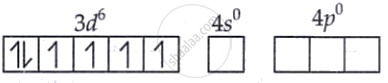
The Co3+ ion has four unpaired electrons in its 3d subshell, and because cobalt is in the +3 oxidation state, NH3 acts as a strong ligand. As a result, the electrons in the 3d orbitals pair up.
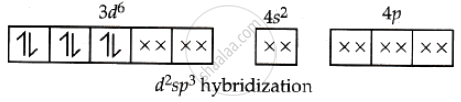
Hence, Co3+ in [Co(NH3)6]3+ forms a d2sp3 hybridised (inner orbital) octahedral complex and is diamagnetic due to the absence of unpaired electrons.