Advertisements
Advertisements
Question
The value of the spin only magnetic moment for one of the following configurations is 2.84 BM. The correct one is:
Options
d5 (in strong ligand field)
d3 (in weak as well as in strong fields)
d4 (in weak ligand fields)
d4 (in strong ligand fields)
Advertisements
Solution
d4 (in strong ligand fields)
Explanation:
(1) d5 - in strong ligand field
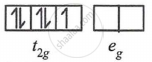
μ = `sqrt("n"("n" + 2))`
= `sqrt3`
= 1.73 BM
(2) d3 - in weak as well as in strong fields
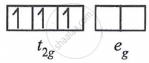
μ = `sqrt(3(5))`
= `sqrt15`
= 3.87 BM
(3) d4 - in weak ligand fields
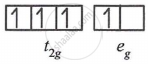
μ = `sqrt(4(4 + 2))`
= `sqrt24`
= 4.89 BM
(4) d4 - in strong ligand fields
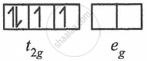
μ = `sqrt(2(4))`
= `sqrt8`
= 2.82 BM
In a strong ligand field, d4 results in 2 unpaired electrons due to pairing in the lower t2g orbitals, giving a magnetic moment of 2.84 BM.
Other configurations either have different numbers of unpaired electrons or lead to different magnetic moment values.
