Advertisements
Advertisements
Question
The total number of possible isomers for [Pt(NH3)4Cl2]Br2 is ______ (nearest integer).
Fill in the Blanks
Advertisements
Solution
The total number of possible isomers for [Pt(NH3)4Cl2]Br2 is 6 (nearest integer).
Explanation:
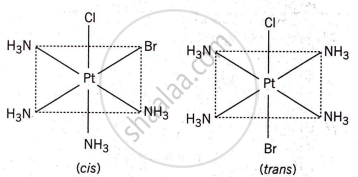
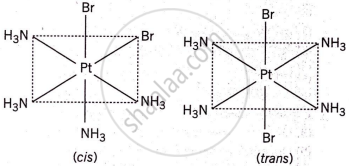
The complex [Pt(NH3)4Cl2]Br2 can have 6 isomers due to a combination of geometrical isomerism and ionization isomerism:
- Geometrical isomerism: The two chloride ions (Cl−) can be arranged in two possible ways around the central platinum ion. (cis and trans)
- Ionization isomerism: The two Br− ions can either be outside or inside the coordination sphere, leading to different isomers.
Thus, combining cis-trans isomerism and ionization isomerism, there are 6 possible isomers.

shaalaa.com
Is there an error in this question or solution?
