Advertisements
Advertisements
Question
The spin-only magnetic moment value for the complex [Co(CN6)]4- is ______ BM. [At. no. of Co = 27]
Options
3.69
2.05
4
1.73
MCQ
Fill in the Blanks
Advertisements
Solution
The spin-only magnetic moment value for the complex [Co(CN6)]4- is 1.73 BM.
Explanation:
[Co(CN6)]4-
x + 6 × (- 1) = - 4
x = ± 2
CO2+ : [Ar] 3d7
and CN- is a strong field ligand which can pair electrons of the central atom.
Transference of electrons
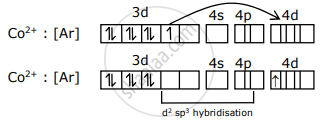
It has one unpaired electron (n) in the 4d - subshell. So spin only magnetic moment (μ) = `sqrt("n"("n + 2"))` BM
where n = number of unpaired electrons.
`mu = sqrt3` BM
μ = 1.73 BM
shaalaa.com
Magnetic Moment of a Coil
Is there an error in this question or solution?
