Advertisements
Advertisements
Question
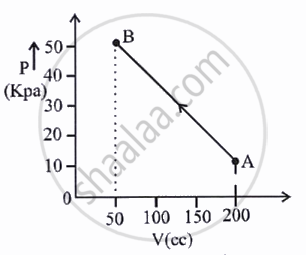
The pressure of a gas changes linearly with volume from A to B as shown in figure. If no heat is supplied to or extracted from the gas then change in the internal energy of the gas will be:
Options
6 J
Zero
−4.5 J
4.5 J
MCQ
Advertisements
Solution
4.5 J
Explanation:
|Δ W| = Area under P-V diagram
= `1/2 xx (50 + 10) xx 10^3 xx 150 xx 10^-6`
= `1/2 xx 60 xx 10^3 xx 150 xx 10^-6`
= 30 × 150 × 10−3
= 4500 × 10−3
4.5 J
As volume is decreasing.
So Δ W = − 4.5 J
By the first law of thermodynamics.
Δ Q = Δ U + Δ W
0 = Δ U − 4.5
Δ U = 4.5 J
shaalaa.com
Is there an error in this question or solution?
