Advertisements
Advertisements
Question
The magnetic moment of central atom of [Co(NH3)6]3+ is ______.
Options
0
1
2
3
MCQ
Fill in the Blanks
Advertisements
Solution
The magnetic moment of central atom of [Co(NH3)6]3+ is 0.
Explanation:
Co = 27 = [Ar] 3d7 4s2
Oxidation state: +3
Co3+ = 24 [Ar] 3d6 4s0
| Orbitals of Co3+ ion | 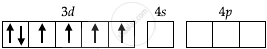 |
| [Co(NH3)6]3+ | 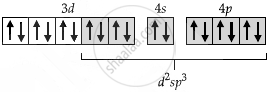 |
| Geometry shape | Octahedral |
| Type of hybridization | d2sp3 |
| No. of unpaired electrons (n) | 0 |
| Magnetic nature | Diamagnetic |
| Nature of complex | Low spin complex, inner orbital complexes |
| Magnetic moment calculation µ = `sqrt("n"("n" + 2))` |
0 |
shaalaa.com
Magnetic Moment of a Coil
Is there an error in this question or solution?
