Advertisements
Advertisements
Question
The elements of 3d transition series are given as:
Sc, Ti, V, Cr, Mn, Fe, Co, Ni, Cu, Zn
Answer the following:
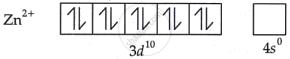
Zn2+ salts are colourless. Why?
Very Short Answer
Advertisements
Solution
The configuration of zinc in its +2 state is [Ar]3d104s0, meaning that every electron is coupled in d orbitals. As a result, its salts are colorless, and the d-d transition is prevented.
shaalaa.com
Is there an error in this question or solution?
2024-2025 (March) Delhi Set 1
