Advertisements
Advertisements
Question
The diagram given below shows the electroplating of a spoon with silver. With reference to the diagram, answer the following questions:
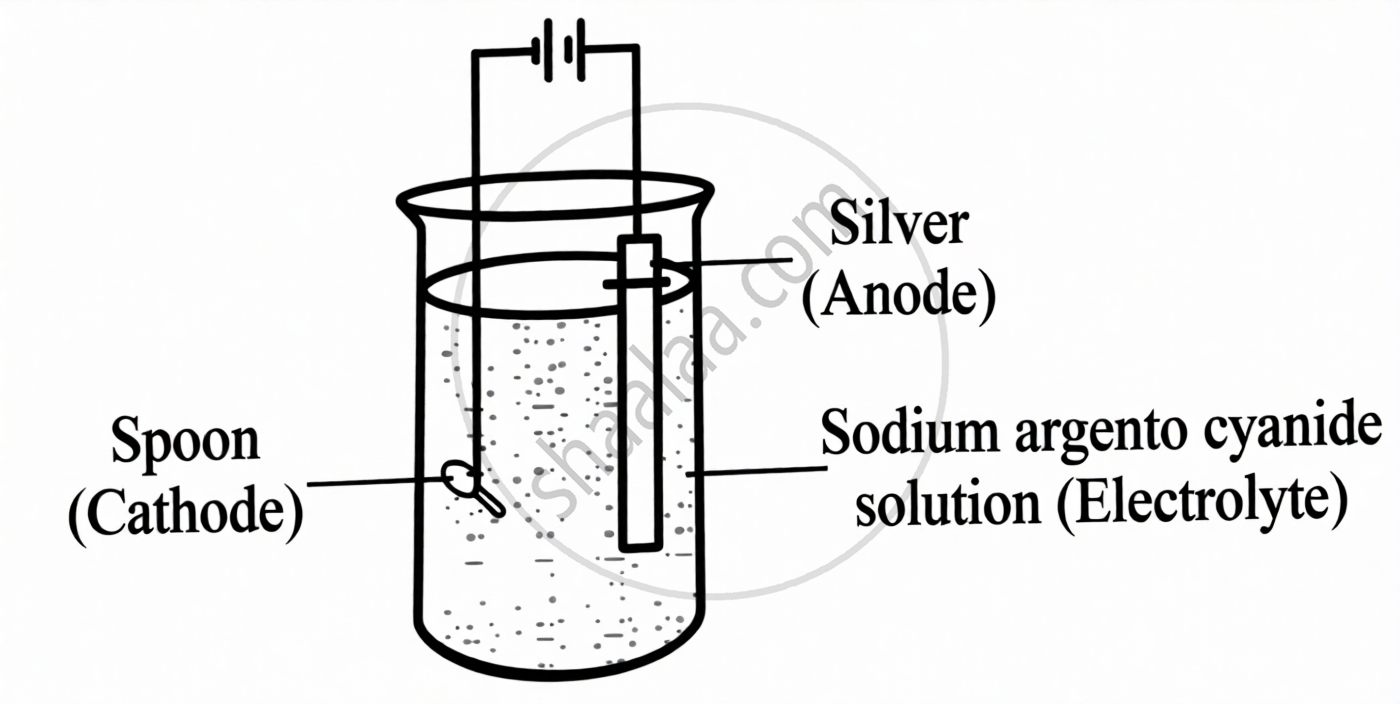
- Write an equation for the reaction that occurs at the cathode.
- Silver nitrate solution is not preferred as an electrolyte in the above process. Give a reason.
- Why is alternating current not used in the process?
- What is the observation at the anode?
Advertisements
Solution
(a) At the cathode (the spoon), silver ions from the electrolyte gain electrons (reduction) to deposit as metallic silver.
\[\ce{Ag^+ + e^- -> Ag}\]
(b) Silver nitrate (AgNO3) is a strong electrolyte that dissociates very rapidly. This would cause rapid, irregular deposition of silver, resulting in a rough, brittle, and nonuniform coating. Using sodium argento cyanide ensures a slow, smooth, and firm finish on the spoon.
(c) Electroplating needs the current to flow in one direction (Direct Current or DC). If you use AC, the current constantly changes direction. This would cause the silver to deposit on the spoon for one second and then dissolve right back off the next second, resulting in no plating at all.
(d) The anode is a block of pure silver that supplies new ions to the solution. The silver anode gradually diminishes in size (wastes away) as it dissolves, replacing the silver ions lost from the electrolyte.
