Advertisements
Advertisements
Question
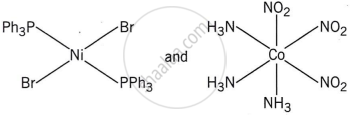
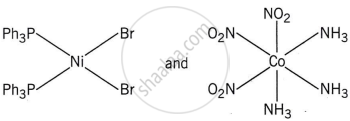
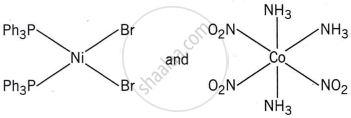
The correct structures of trans-[NiBr2(PPh3)2] and meridonial- [Co(NH3)3(NO2)3] respectively are:
Options
MCQ
Advertisements
Solution
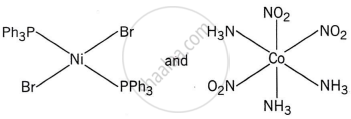
Explanation:
The complex trans-[NiBr2(PPh3)2] has a square planar geometry, with the trans arrangement placing the two Br ligands opposite each other and the two PPh3 ligands opposite each other as well.
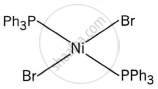
Meridonial-[Co(NH3)3(NO2)3] is the meridional isomer, where the three NO2 ligands are arranged adjacent to each other in an octahedral geometry.
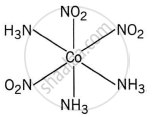
shaalaa.com
Is there an error in this question or solution?