Advertisements
Advertisements
Question
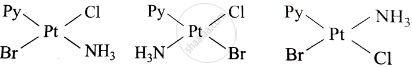
The complex [(Pt(Py)(NH3)BrCl] will have how many geometrical isomers?
Options
3
4
0
2
MCQ
Advertisements
Solution
3
Explanation:
The complex [(Pt(Py)(NH3)BrCl)] has 3 geometrical isomers because of the following:
- Cis-trans isomerism between Br and Cl (adjacent or opposite).
- Py and NH3 can switch positions, adding another isomer.
shaalaa.com
Isomerism in Coordination Compounds - Stereoisomerism
Is there an error in this question or solution?
