Advertisements
Advertisements
Question
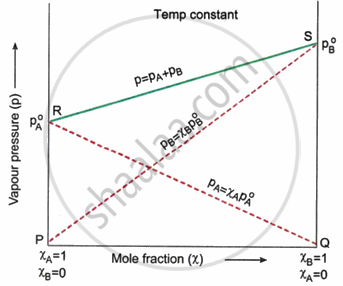
State and explain Raoult’s law. Show graphically the variation of total vapour pressure over a mixture of two volatile liquids with the composition of the mixture.
Advertisements
Solution
Raoult’s Law states that for a solution of two or more miscible volatile liquids, the partial vapour pressure of each component of the solution at a given temperature is directly proportional to its mole fraction in the solution.
Mathematically, it can be expressed as
`p_A = p_A^circ * chi_A`
`p_B = p_B^circ * chi_B`
Where
pA and pB are the partial vapour pressures of components A and B, respectively.
`p_A^circ` and `p_B^circ` are the vapor pressures of the pure components A and B at the same temperature.
χA and χB are the mole fractions of components A and B, respectively.
Two volatile liquids, A and B, exert their own vapour pressures above the solution when they are combined. The sum of the partial pressures can be used to determine the total vapour pressure P above the solution.