Advertisements
Advertisements
Question
Spin only magnetic moment in BM of \[\ce{[Fe(CO)4(C2O4)]+}\] is ______.
Options
5.92
0
1
1.73
MCQ
Fill in the Blanks
Advertisements
Solution
Spin only magnetic moment in BM of \[\ce{[Fe(CO)4(C2O4)]+}\] is 1.73.
Explanation:
\[\ce{[Fe(CO)4(C2O4)]+}\]
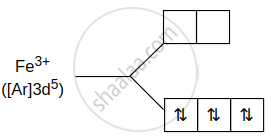
CO is strong field ligand, so pairing will take place.
∴ Spin only magnetic moment is:
`mu = sqrt("n"("n + 2"))` = 1 (1 + 2) = 1.73 B. M.
shaalaa.com
Magnetic Moment of a Coil
Is there an error in this question or solution?
