Advertisements
Advertisements
Question
Solubility curve of a hydrated salt in water with temperature is given. The curve indicates that the solution process is: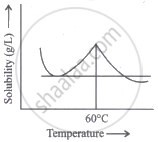
Options
Exothermic
Endothermic
Endothermic till 60°C and endothermic after 60°C
Endothermic till 60°C and exothermic thereafter
MCQ
Advertisements
Solution
Endothermic till 60°C and exothermic thereafter
Explanation:
The solubility curve of a hydrated salt in water exhibits an endothermic response up to 60°C and an exothermic reaction after that.
shaalaa.com
Calorimetric Measurement of ΔU and ΔH
Is there an error in this question or solution?
