Advertisements
Advertisements
Question
Show that the packing fraction in a simple cubic unit cell is 0.524 and the radius of the atom is equal to half of the edge length.
Numerical
Advertisements
Solution
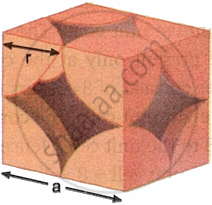
A simple cubic unit cell contains only one atom. Suppose the edge length of a simple cubic unit cell is a, and the atom’s radius is r.
In a simple cubic unit cell, the two spheres at the cube’s corners touch, as seen in Fig.
a = 2r
Number of atoms per unit cell = `8 xx 1/8 = 1`
Volume of an atom = `4/3 pi r^3`
Volume of unit cell (cube) = a3
Volume of unit cell (cube) = (2)3
Volume of unit cell (cube) = 8r3
∴ Packing fraction = `"Volume occupied by atoms"/"Volume of unit cell"`
= `((4/3) pi r^3)/(8 r^3)`
= 0.524
shaalaa.com
Is there an error in this question or solution?
