Advertisements
Advertisements
Questions
PH3 forms bubbles when passed slowly in water but NH3 dissolves. Explain why?
On being slowly passed through water, PH3 forms bubbles but NH3 dissolves. Why is it so?
Advertisements
Solution 1
In PCl5, phosphorus undergoes sp3d hybridisation, and a trigonal bipyramidal configuration comes into existence.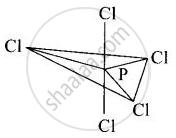
The equatorial P-Cl bonds are equivalent, while two axial bonds are different and larger than equatorial bonds.
Solution 2
- Ammonia (NH3) dissolves in water because it can form hydrogen bonds with water molecules due to the presence of a lone pair on nitrogen and N-H bonds.
- Phosphine (PH3) forms bubbles when passed through water because it cannot form hydrogen bonds with water; its bonds are weaker and mainly van der Waals forces.
- Hence, NH3 is highly soluble in water, while PH3 remains largely insoluble and escapes as bubbles.
This difference arises from the ability of NH3 to hydrogen bond, but PH3 cannot.
APPEARS IN
RELATED QUESTIONS
Account for the following : Solid PCl5 is ionic in nature.
Complete the following equations: Ag+PCl5 →
What happens when PCl5 is heated?
Write a balanced equation for the hydrolytic reaction of PCl5 in heavy water.
Can PCl5 act as an oxidising as well as a reducing agent? Justify.
Complete and balance the following equation:
\[\ce{PCl3 + H2O ->}\]
PCl5 is possible but NCl5 does not exist:
SF6 is known but SCl6 is not. Why?
On reaction with \[\ce{Cl2}\], phosphorus forms two types of halides ‘A’ and ‘B’. Halide A is yellowish-white powder but halide ‘B’ is colourless oily liquid. Identify A and B and write the formulas of their hydrolysis products.
White phosphorus reacts with chlorine and the product hydrolyses in the presence of water. Calculate the mass of \[\ce{HCl}\] obtained by the hydrolysis of the product formed by the reaction of 62 g of white phosphorus with chlorine in the presence of water.
What is the smoke screen?
