Advertisements
Advertisements
Question
One mole of an ideal diatomic gas undergoes a transition from A to B along a path AB as shown in the figure,
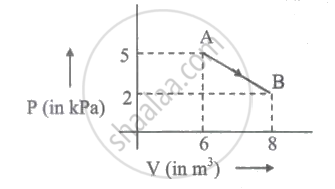
The change in internal energy of the gas during the transition is ____________.
Options
35 k J
- 35 k J
20 J
- 20 k J
MCQ
Fill in the Blanks
Advertisements
Solution
The change in internal energy of the gas during the transition is - 35 k J.
Explanation:
Change in internal energy,
`Delta "U" = ("P"_2 "V"_2 - "P"_1 "V"_2)/(gamma - 1)`
`= (2 xx 8 - 5 xx 6)/(7/5 -1) .......(becausegamma = 7/5 "for ideal diatomic gas") = -35 "k J"`
shaalaa.com
Sterling Cycle
Is there an error in this question or solution?
