Advertisements
Advertisements
Question
On heating lead (II) nitrate gives a brown gas “A”. The gas “A” on cooling changes to colourless solid “B”. Solid “B” on heating with NO changes to a blue solid ‘C’. Identify ‘A’, ‘B’ and ‘C’ and also write reactions involved and draw the structures of ‘B’ and ‘C’.
Advertisements
Solution
The gas A is \[\ce{NO2}\]. The reactions are explained as under:
\[\ce{2Pb(NO3)2 ->[Heat][673 K] + 2PbO + \underset{(Brown colour)}{\underset{Nitrogen dioxide}{\underset{(A)}{4NO2}}} + O2}\]
\[\ce{2NO2 ->[Cooling] \underset{Solid (Colourless)}{\underset{(B)}{N2O4}}}\]
\[\ce{2NO + N2O4 ->[Heating] \underset{Dinitrogen trioxide}{\underset{(C)}{\underset{Blue solid}{2N2O3}}}}\]
Structure of B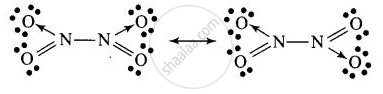
Structure of C
APPEARS IN
RELATED QUESTIONS
Write balanced chemical equations for action of potassium permanganate on: Hydrogen
Pb(NO3)2 on heating gives a brown gas which undergoes dimerisation on cooling? Identify the gas
Why does NO2 dimerise?
Why does the reactivity of nitrogen differ from phosphorus?
Give the resonating structure of N2O5.
Which of the following is a basic oxide?
(A) SiO2
(B) P4O10
(C) MgO
(D) Al2O3
Explain why Mn2+ ion is more stable than Mn3+? (Given : Mn→ Z=25)
Account for the following :
Bond angle in NH+4NH4+ is higher than NH3.
Tendency to show –3 oxidation state decrease from Nitrogn (N) to Bismuth (Bi).
Which of the following statements is not correct for nitrogen?
Pick out the wrong statement.
Pure nitrogen is prepared in the laboratory by heating a mixture of:
Which of the following oxides is neutral?
The bonds present in N2O5 are ____________.
Which of the following oxides of nitrogen is a coloured gas?
A deep brown gas is formed by mixing two colourless gases which are:
Which of the following elements can be involved in pπ – dπ bonding?
Assertion: \[\ce{HNO3}\] makes iron passive.
Reason: \[\ce{HNO3}\] forms a protective layer of ferric nitrate on the surface of iron.
Heating white phosphorus with conc. NaOH solution gives mainly ______.
