Advertisements
Advertisements
Question
Observe the figure and answer the following questions:
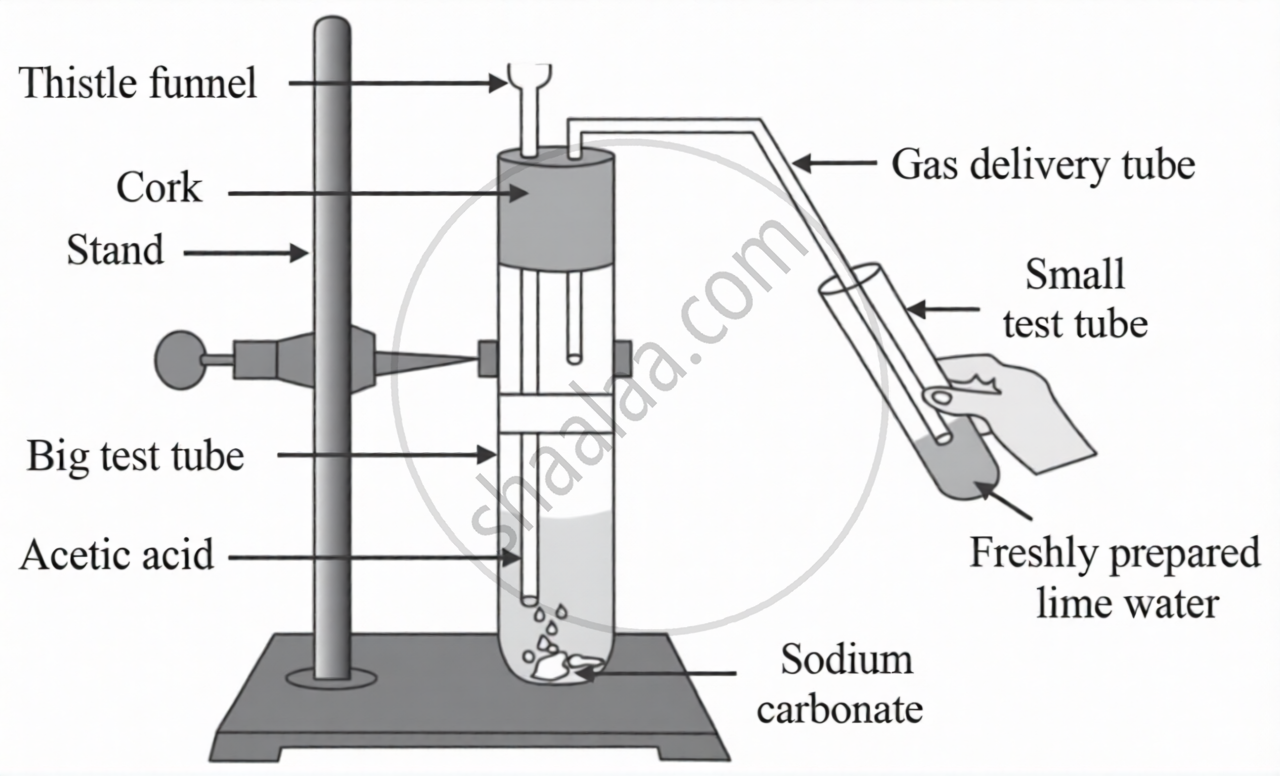
- Which gas is released out as effervescence in the big test tube?
- What is the colour change in the lime water?
- Write the related chemical equation.
Chemical Equations/Structures
Short Answer
Advertisements
Solution
- The gas released with effervescence in the big test tube is carbon dioxide (CO2).
- The lime water turns milky (white) due to the formation of calcium carbonate.
- The related chemical equation is:
\[\ce{\underset{Acetic acid}{2CH3COOH} + \underset{Sodium carbonate}{Na2CO3} -> \underset{Sodium acetate}{2CH3COONa} + \underset{Water}{H2O} + \underset{Carbon dioxide}{CO2}}\]
shaalaa.com
Is there an error in this question or solution?
