Advertisements
Advertisements
Question
Nickel (Z = 28) combines with a uninegative monodentate ligand X− to form a paramagnetic complex [NiX4]2−. The number of unpaired electron(s) in the nickel and geometry of this complex ion respectively are ______.
Options
one, tetrahedral
two, tetrahedral
one, square planar
two, square planar
MCQ
Fill in the Blanks
Advertisements
Solution
Nickel (Z = 28) combines with a uninegative monodentate ligand X− to form a paramagnetic complex [NiX4]2−. The number of unpaired electron(s) in the nickel and geometry of this complex ion respectively are two, tetrahedral.
Explanation:
The formation and geometry of [NiX4]2− are as follows:
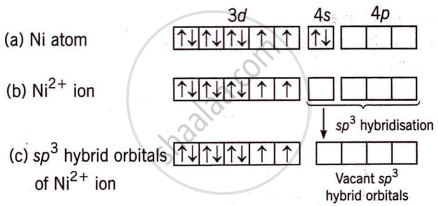
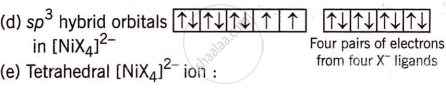
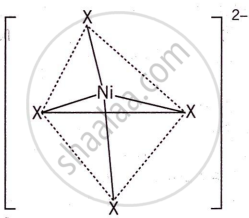
The complex has a tetrahedral shape and two unpaired electrons, making it paramagnetic.
shaalaa.com
Is there an error in this question or solution?
