Advertisements
Advertisements
Question
Name the following:
What are the main natural sources of alkane?
Advertisements
Solution
Crude petroleum and natural gas.
APPEARS IN
RELATED QUESTIONS
Choose the correct option.
Acidic hydrogen is present in _______.
Choose the correct option.
1-Chloropropane on treatment with alc. KOH produces _______.
Name the following:
The type of hydrocarbon that is used as a lubricant.
Write a balanced chemical reaction for the preparation of ethane from Ethyl magnesium iodide.
How many monochlorination product is possible for 2-methylpropane? Draw their structures and write their IUPAC names.
What is the action of following on ethyl iodide?
Zn, HCl
An alkene ‘A’ on ozonolysis gives 2 moles of ethanal. Write the structure and IUPAC name of ‘A’.
\[\ce{C2H5Br + 2Na ->[dry ether]C4H10 + 2NaBr}\]
The above reaction is an example of which of the following.
In the following reaction,
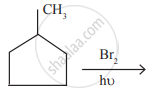
The major product obtained is
The general formula for cyclo alkanes.
\[\begin{array}{cc}
\ce{CH2 - CH2 ->[(A)] CH ≡ CH}\\
|\phantom{.......}|\phantom{.................}\\
\ce{Br}\phantom{.....}\ce{Br}\phantom{.................}
\end{array}\], where A is,
The molecular formula of an alkane with molecular weight 86 is ____________.
Which of the following undergoes substitution reaction?
Which of the following alcohols is prepared by acid catalyzed hydration of alkenes?
When HCl is treated with propene in presence of sodium peroxide, the major product obtained is ____________.
Arrange the three isomers of alkane with malecular formula C5 H12 in increasing order of boiling points and write their IUPAC names.
Arrange the three isomers of alkane with malecular formula C5H12 in increasing order of boiling points and write their IUPAC names.
Write the balanced chemical reaction for the preparation of ethane from Ethyl magnesium iodide
Write the balanced chemical reaction for preparation of ethane from Ethyl bromide.
Write the balanced chemical reaction for Preparation of ethane from Ethyl bromide.
Acetone and acetaldehyde are the ozonolysis products of an alkene. Write the structural formula of an alkene and give IUPAC name of it.
Write the balanced chemical reaction for Preparation of ethane from Ethyl magnesium iodide.
Write the balanced chemical reaction for preparation of ethane from Ethyl bromide
Write a balanced chemical reaction for the preparation of ethane from Ethyl bromide.
Arrange the three isomers of alkane with malecular formula \[\ce{C5H12}\] in increasing order of boiling points and write their IUPAC names.
Write the balanced chemical reaction for preparation of ethane from Ethyl magnesium iodide.
Write the balanced chemical reaction for preparation of ethane from Ethyl magnesium iodide.
How many monochlorination product is possible for 2-methylpropane? Draw the structure and write the IUPAC name.
Write the balanced chemical reaction for preparation of ethane from Ethyl bromide.
