Advertisements
Advertisements
Question
Lemon juice and vinegar taste ______ because they contain ______.
Advertisements
Solution
Lemon juice and vinegar taste sour because they contain acids.
APPEARS IN
RELATED QUESTIONS
Which gas is usually liberated when an acid reacts with a metal? How will you test for the presence of this gas?
What happens to temperature of the solution, when an acid is neutralised?
Bee sting contains:
(a) an acidic liquid
(b) a salt solution
(c) an alkaline liquid
(d) an alcohol
Give one example in the following case:
A hydrogen containing compound which is not an acid.
If iron reacts with dilute sulphuric acid, what will be the products?
Complete the following table and write one equation for each to justify the statement:
|
Reactants |
Products |
Method |
|
Soluble base + Acid (dil) |
Salt + water |
Neutralisation Titration |
|
Metal + Non-metal |
Salt (soluble/insoluble) |
______ |
|
Insoluble base + ______ |
Salt (soluble) + water |
______ |
|
Active metal + Acid (dil) |
______ + ______ |
______ |
|
Soluble salt solution (A) + Soluble salt solution (B) |
Precipitated salt + Soluble salt |
______ |
|
Carbonate/bicarbonate + Acid (dil) |
Salt + ______ + ______ |
Decomposition of carbonate |
|
Chlorides/nitrates + Acid (conc.) |
______ + ______ |
Decomposition of chlorides and nitrates |
Answer the following question.
Identify the acid and the base from which sodium chloride is obtained. Which type of salt is it? When is it called rock salt? How is rock salt formed?
What happens when a solution of an acid is mixed with a solution of a base in a test tube?
(i) Temperature of the solution decreases.
(ii) Temperature of the solution increases.
(iii) Temperature of the solution remains the same.
(iv) Salt formation takes place.
Fill in the cross word given in figure with the help of the clues provided.
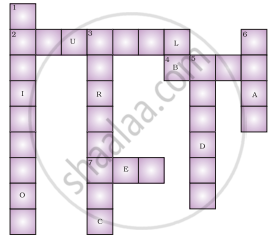
Across
(2) The solution which does not change the colour of either red or blue litmus.
(4) Phenolphthalein gives pink colour in this type of solution.
(7) Colour of blue litmus in lemon juice.
Down
(1) It is used to test whether a substance is acidic or basic.
(3) It is a natural indicator and gives pink colour in basic solution.
(5) Nature of ant’s sting.
(6) It is responsible for the increase in temperature during a neutralisation reaction.
Complete the following equation.
Mg+ ______ → ______ + H2.
