Advertisements
Advertisements
Question
Intramolecular hydrogen bonding is present in:
Options
HF
MCQ
Advertisements
Solution

Explanation:
Hydrogen bonding occurs within a single molecule. This happens when a molecule contains two functional groups.
HF allows for intermolecular hydrogen bonding but not intramolecular bonding.
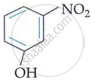
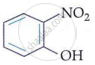
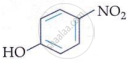
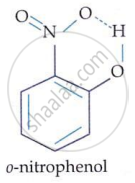
In m-nitrophenol and p-nitrophenol, the −NO2 and −OH groups are widely apart, preventing intramolecular hydrogen bonding. In o-nitrophenol, the −NO2 and −OH groups on neighbouring carbon atoms generate intramolecular hydrogen bonding.
shaalaa.com
Is there an error in this question or solution?
