Advertisements
Advertisements
Question
In the following diagram, if acetic acid of same concentration is taken in place of dilute sulphuric acid, then:
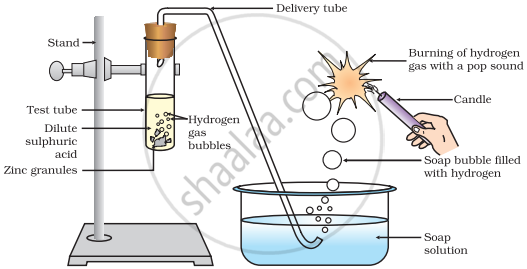
Options
Same amount of H2 gas will be evolved.
H2 gas will not be evolved.
The amount of H2 gas evolved in less amount.
In place of H2 gas, O2 gas will be evolved.
MCQ
Advertisements
Solution
The amount of H2 gas evolved in less amount.
Explanation:
Dilute sulphuric acid is a strong acid and ionizes completely in water. Acetic acid is a weak acid and ionizes only partially. Fewer H+ ions are available in acetic acid compared to sulphuric acid. Hence, less hydrogen gas will be produced when acetic acid is used. Since acetic acid is a weak acid, the amount of hydrogen gas evolved will be less compared to dilute sulphuric acid.
shaalaa.com
Is there an error in this question or solution?
