Advertisements
Advertisements
Question
If ‘X’ g. of potassium nitrate is added to 100 g. of water at 60°C and the salt dissolves completely then :
(a) is ‘X’ g. the solubility of potassium nitrate at 60°C.
(b) is the solution formed – saturated or unsaturated
(c) if on addition ‘X’ + ‘Y’ g. of potassium nitrate to the same amount of water at the same temperature and the solute now just remains behind after stirring then –
(d) is the solution now – saturated or unsaturated
(e) is ‘X’ + ‘Y’ g. the solubility of potassium nitrate.
Advertisements
Solution
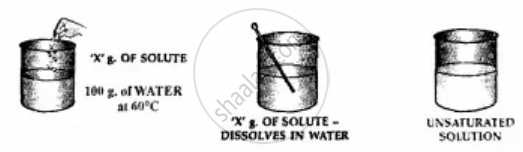
Add ‘X’ g. of solute i.e. potassium nitrate to 100 g. of water 60°C.
1. Stir the solute i.e. potassium nitrate in water thoroughly.
2. ‘X’ g. of the solute completely dissolves in water.
3. Add more solute and again stir thoroughly.
4. The solute continues to dissolves.
5. Water i.e. the solvent can dissolve more of the solute at the given temperature.
6. The solution is, therefore, is said to be unsaturated.
Add more solute to water till on adding an amount ‘X + Y’ g. of the solute i.e. potassium nitrate to 100 g. of water at 60°C.
1. The solute just remains behind after stirring.
2. The solution is now saturated.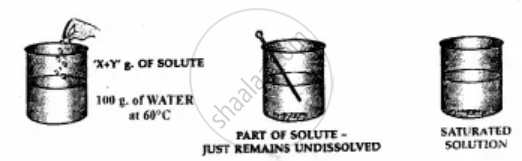
A saturated solution cannot dissolve more of the solute at a given temperature.
RELATED QUESTIONS
Define Solvent.
State two factors by which solubility of a solute in a solvent can be increased.
Name the following
The liquid or medium of dissolution which allows the solute to dissolve in it.
State whether the following statement is true or false. If false write the correct statement.
Solubility of most solids – decreases with increase in temperature
Define the term solute.
State the characteristics of a true solution.
Differentiate between the following:
Solute and solvent – forming a solution.
What is a solution?
Polar compounds are soluble in _____ solvents
A homogeneous mixture of two or more substances is called ______.
