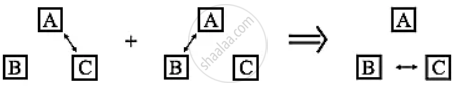
- The figure shows a schematic representation of the zeroth law of thermodynamics.
- The double arrow represents thermal equilibrium between systems.
- If systems A and C are in thermal equilibrium, and systems A and B are in thermal equilibrium, then systems B and C must be in thermal equilibrium.
- Then it can be inferred that systems A, B and C are at the same temperature.
Advertisements
Advertisements
Question
“If two systems are each in thermal equilibrium with a third system, they are also in thermal equilibrium with each other.” This statement refers to ______.
Options
zeroth law of thermodynamics
first law of thermodynamics
second law of thermodynamics
Carnot’s law
MCQ
Fill in the Blanks
Advertisements
Solution
“If two systems are each in thermal equilibrium with a third system, they are also in thermal equilibrium with each other.” This statement refers to the zeroth law of thermodynamics.
Explanation:
shaalaa.com
Is there an error in this question or solution?
