Advertisements
Advertisements
Question
If ΔTb is the elevation of boiling point and m is the molality of solution, show that ΔTb = Kbm.
Advertisements
Solution
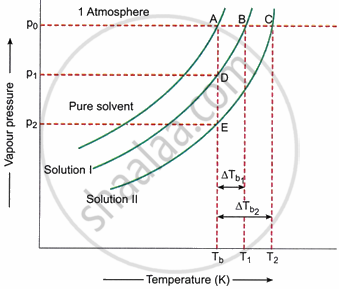
In Fig. if curves BD and CE are assumed to be straight lines, triangles ABD and ACE may be regarded as similar triangles.
For similar triangles ABD and ACE, we have
`(A B)/(A C) = (A D)/(A E)`
or `(T_1 - T_b)/(T_2 - T_b) = (p^circ - p_1)/(p^circ - p_2)` ...(i)
Where, p° = Vapour pressure of pure solvent at its boiling point Tb (equal to 1 atm)
p1 = Vapour pressure of solution I at temperature Tb
p2 = Vapour pressure of solution II at temperature Tb.
Eq. (i) can be written as
`(Delta T_(b_1))/(Delta T_(b_2))` ...(ii)
Where, `Delta T_(b_1)` = Elevation of boiling point for solution I
`Delta T_(b_2)` = Elevation of boiling point for solution II
Δp1 = Lowering in vapour pressure for solution I
Δp2 = Lowering in vapour pressure for solution II.
It follows from eq. (ii) that in general,
ΔTb ∝ Δp ...(iii)
i.e., the elevation of boiling point is directly proportional to the lowering in vapour pressure.
According to Raoult’s law, for a dilute solution,
`(p^circ - p)/p^circ = (w M)/(W M^')`
or `(Delta p)/p^circ = (w M)/(W M^')`
or `Delta p = p^circ M xx w/(W M^')`
Since p°M is a constant for a particular solvent, we have
`Delta p prop w/(W M^')` ...(iv)
Comparing eqs. (iii) and (iv), we have
`Delta T_b prop w/(W M^')` ...(v)
If W (mass of solvent) = 1 kg, `w/(W M^')` represents the molality m of the solution. Therefore,
ΔTb ∝ m
or ΔTb = Kb . m
