Advertisements
Advertisements
Question
Identify the geometry of TeF4 molecule from the following.
Options
Linear
Tetrahedral
Trigonal bipyramidal
Trigonal
MCQ
Advertisements
Solution
Trigonal bipyramidal
Explanation:
Te is from group 16, which means it has 6 electrons in valence shell out of which 4 are bonded with 1 Fluorine atom each thus leaving behind 1 lone pair.
TeF4 has 1 lone pair and 4 bonding pairs (AB4E). Its electron geometry is trigonal bipyramidal.
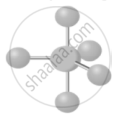
shaalaa.com
Is there an error in this question or solution?
