Advertisements
Advertisements
Question
Identify the correct answer.
Options
Dipole moment of NF3 is greater than that of NH3.
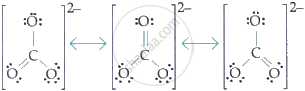
Three canonical forms can be drawn for \[\ce{CO^{2-}_3}\] ion.
Three resonance structures can be drawn for ozone.
BF3 has non-zero dipole moment.
MCQ
Advertisements
Solution
Three canonical forms can be drawn for \[\ce{\mathbf{CO^{2-}_3}}\] ion.
Explanation:
The canonical forms can be drawn from the \[\ce{CO^{2-}_3}\].
shaalaa.com
Is there an error in this question or solution?
