Advertisements
Advertisements
Question
Identify the substance underlined, in the following case:
Cation that does not form a precipitate with ammonium hydroxide but forms one with sodium hydroxide.
Advertisements
Solution
Cation that does not form a precipitate with ammonium hydroxide but forms one with sodium hydroxide: Ca2+
APPEARS IN
RELATED QUESTIONS
Choose the correct word or phrase from the bracket to complete the following sentence:
Heating ammonium chloride with sodium hydroxide produces ______.
Choose the correct word or phrase from the bracket to complete the following sentences.
Heating solution of ammonium chloride with sodium nitrite produces ______
Name the other ion formed when ammonia dissolves in water
The diagram shows an experimental set up for the laboratory preparation of a pungent smelling gas. The gas is alkaline in nature.
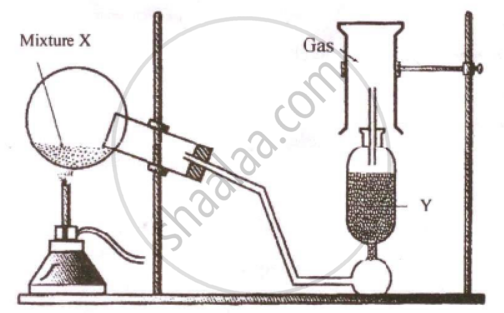
- Nature the gas collected in the jar.
- Write the balance equation for the above preparation.
- How is the gas being collected?
- Name the drying agent used.
- How will you find that the jar is full of gas?
What do you observe when Ammonia is passed over heated lead oxide.
What do you observe when Ammonia solution is added drop by drop and then in excess to aqueous copper sulphate solution.
Differentiate between:
Aqueous ferrous and ferric sulphate solution
Choose the correct answer from the options given below :
Ammonia is produced when ammonium chloride is heated with
Write an equation to illustrate the reducing nature of ammonia.
