Advertisements
Advertisements
Question
Identify the process shown in the figure and name it.
Advertisements
Solution
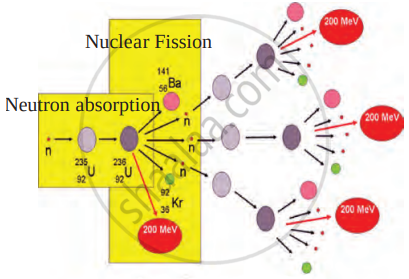
The given figure is of nuclear fission (Chain reaction).
APPEARS IN
RELATED QUESTIONS
Which of the following is not an example of a bio-mass energy source?
Name the type of nuclear reaction which is involved in the working of:
a hydrogen bomb.
State one use of nuclear fission reactions.
Fill in the following blank with suitable word:
Nuclear .......... is used in nuclear power stations for the production of electricity.
What is a nuclear reactor? What is the fuel used in a nuclear reactor?
One type of energy which has not been controlled so far is:
(a) ocean thermal energy
(b) nuclear fusion energy
(c) geothermal energy
(d) nuclear fission energy
Which of the following can undergo nuclear fusion reaction?
(a) uranium
(b) deuterium
(c) barium
(d) krypton
The mass numbers of four elements A, B, C and D are 2, 20, 135 and 235, respectively. Which one of them will be most suitable to make:
a hydrogen bomb?
Explain with a diagram the step-by-step energy conversion in a nuclear power plant.
In what way are these numbers related to the isotopes of an element?
Write down the Einstein's mass-energy equivalence relation, explaining the meaning of each symbol used in it.
Complete the following nuclear change:
\[\ce{^24_11 Na -> ...Mg.... + ^0_-1β}\]
State, giving reason, whether the following nuclear decay are allowed or not?
\[\ce{_Z^AX ->_Z^{A-1}Y + _-1^0e}\]
The nucleus 84X202emits an alpha particle and forms the nucleus Y. Represent this change in the form of an equation.
When an alpha particle gains two electrons it becomes neutral and becomes an atom of an element which is a rare gas. What is the name of this rare gas?
Define.
Nuclear fission
Write the demerits of a nuclear energy plant.
What is stellar energy?
Give scientific reasons.
It is absolutely necessary to control the fission reaction in nuclear power plants.
