Advertisements
Advertisements
Question
Identify a more favourable resonance structure from the following. Justify.

One Line Answer
Advertisements
Solution
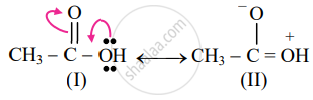
Structure (I) will be a more favourable resonance structure as structure (II) involves the separation of opposite charges and the electronegative oxygen atom has a positive charge.
shaalaa.com
Is there an error in this question or solution?
Chapter 14: Basic Principles of Organic Chemistry - Exercises [Page 230]
