Advertisements
Advertisements
Questions
How would you account for the increase in the reaction rate on increasing the temperature on the basis of collision theory?
How does the collision theory explain the effect of temperature on the reaction rates?
Advertisements
Solution
Collision theory states that not all molecular collisions are efficient and do not produce a chemical reaction that produces products: The creation of products is only caused by effective collisions, or collisions between molecules with enough energy to cross the energy barrier and with the right orientation. As can be seen from the figure, the number of such molecules makes up a very small portion of all molecules. This explains why a chemical reaction occurs in a small number of collisions.
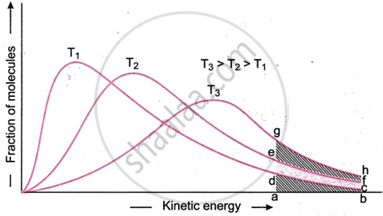
Distribution of energy among molecules at three temperatures-T1, T2 and T3.
The energy distribution curves at three temperatures, T1, T2, and T3, are displayed in the figure so that T3 > T2 > T1. Some molecules have relatively high kinetic energy at temperature T1, while others have minimal kinetic energy. The majority of the molecules do, however, have intermediate kinetic energies. The percentage of molecules that are contained inside the shaded area of the abed has kinetic energy that is higher than or equal to the threshold energy and can go through effective conditions.
The whole curve is to higher energies as the temperature rises from T1 to T2. As a result, the percentage of molecules with energy above or equal to the threshold energy rises. The area efcd in indicates this rise. figure. Currently, the region efba contains the whole proportion of molecules with energy greater than or equal to the threshold energy at temperature T2. This fraction (shown by area abcd) is much more than that at temperature T1.
Therefore, more molecules can experience effective collisions at temperature T2. As a result, the temperature T2 reaction rate will be substantially higher than the temperature T1 reaction rate. The fraction of molecules with energy greater than or equal to the threshold energy grows further if the temperature is raised to T3 (the figure shows this increase as area ghfe). The rate of reaction rises further as a result of a further increase in the fraction of molecules that can experience effective collisions.
Hence, it may be concluded that an increase in temperature increases the number of effective collisions, resulting in an increase in the rate of reaction.
Even with a small rise in temperature, the number of molecules with energy greater than or equal to the threshold energy rises significantly. As a result, even a slight increase in temperature causes a noticeable increase in a reaction's flow. In the majority of processes, the fraction of such molecules increases two to three times when the system’s temperature is raised by 10°. Because of this, the pace of most reactions rises two to three times for every 10° increase in temperature.
