Advertisements
Advertisements
Question
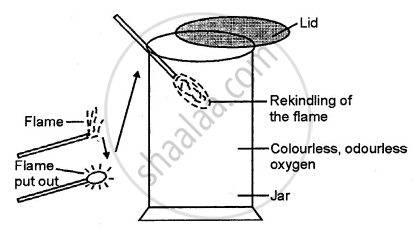
How will you identify oxygen in the laboratory ?
Short/Brief Note
Advertisements
Solution
Oxygen is a colourless and odourless gas which rekindles a glow¬ing splinter.
shaalaa.com
Properties of Oxygen
Is there an error in this question or solution?
APPEARS IN
RELATED QUESTIONS
How is oxygen naturally renewed in air ?
State the use of manganese dioxide in the laboratory prepa-ration of oxygen from potassium chlorate.
How will you obtain dry oxygen in the laboratory?
Complete and balance the following equation
\[\ce{N2 + O2 ->[3000°C]}\]
State any four physical properties of oxygen gas.
Describe what you will observe and write fully balanced equation when the following is introduced in a jar of oxygen.
Burning magnesium
Give tests for oxygen gas.
Oxygen is a poor conductor of heat and electricity.
Write about the combustible property of Oxygen.
Write a note on the rusting process.
