Advertisements
Advertisements
Question
How will you convert the following?
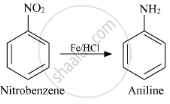
Nitrobenzene into aniline
Advertisements
Solution
Nitrobenzene into aniline
shaalaa.com
Is there an error in this question or solution?
How will you convert the following?
Nitrobenzene into aniline
Nitrobenzene into aniline
