Advertisements
Advertisements
Question
How is zinc extracted from zinc oxide?
Advertisements
Solution
The zinc blende ore is the primary source of zinc. The following procedures are used to perform the extraction.
Concentration: In ball mills, the ore is crushed and ground into a fine powder. Levigation, a gravity technique, is used to concentrate the finely ground ore, followed by froth flotation. The ore undergoes electromagnetic separation if iron oxide is also present.
Roasting: The concentrated ore is roasted at approximately 1200 K with extra air. Zinc sulphide in ore or zinc sulphate produced during oxidation is transformed to zinc oxide.
\[\ce{2ZnS + 3O2 -> 2ZnO + 2SO2}\]
\[\ce{ZnS + 2O2 -> ZnSO4}\]
Calamine ore needs only calcination.
\[\ce{ZnCO3 -> ZnO + CO2}\]
Reduction: Several processes are utilised to reduce the zinc oxide produced in the above stage. The vertical retort process involves mixing roasted ore with powdered coal or coke and heating in a vertical retort.
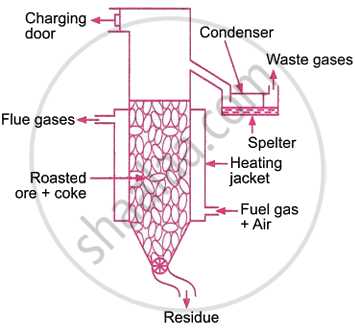
The retort is enclosed by a jacket that burns a mixture of fuel gas and air. The heat produced heats the retort to around 1600 K. Zinc oxide is converted to zinc.
\[\ce{ZnO + C ->[1600 K] Zn + CO}\]
The zinc produced is then vapourised. Zinc and CO vapours escape the retort towards the top and condense. The resulting condensed zinc is known as zinc spelter. Carbon monoxide doesn't condense and escapes. The charging door adds a new charge, while a screw conveyer at the bottom removes any residue. Therefore, the process is continual.
