Advertisements
Advertisements
Question
How is the osmotic pressure of a solution measured?
Very Long Answer
Advertisements
Solution
Several methods are available for the measurement of osmotic pressure of a solution. Among these, the most widely used method is Berkley and Hartley’s method.
- This method involves the measurement of external pressure which is just sufficient to stop the flow of solvent into solution through a semipermeable membrane.
- This pressure is equal to the osmotic pressure of the solution.
- The apparatus used in this method is shown in the figure. It consists of a porcelain tube with copper ferrocyanide precipitate deposited in its walls.
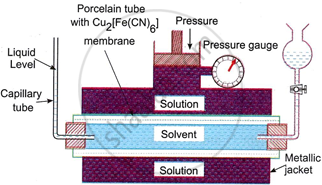
- The copper ferrocyanide precipitate acts as a semipermeable membrane. The porcelain tube is enclosed in a metallic jacket.
- The tube is fitted with a solvent (water) reservoir at one end and a capillary tube at the other end. The metallic jacket is fitted with a piston connected to a pressure gauge to directly measure the pressure applied to the solution.
- The porcelain tube is filled with pure solvent and the metallic jacket is filled with the solution whose osmotic pressure is to be determined.
- As osmosis proceeds, the level of the liquid in the capillary tube tends to go down due to the flow of solvent into the solution through the semipermeable membrane.
- Now with the help of a piston, an external pressure is applied to the solution. The applied pressure should be just sufficient to maintain the level of liquid stationary in the capillary tube.
- At this stage, osmosis is checked and the applied pressure is equal to the osmotic pressure of the solution. It can directly be read on the pressure gauge.
shaalaa.com
Is there an error in this question or solution?
