Advertisements
Advertisements
Question
How is methane prepared in the laboratory?
Chemical Equations/Structures
Diagram
Long Answer
Advertisements
Solution
Reactants: Sodium ethanoate (sodium acetate) and soda lime.
Since sodium hydroxide is deliquescent, soda lime (a mixture of sodium hydroxide and calcium oxide) is used, which is in the form of a white porous mass or granules.
Procedure: A mixture of sodium ethanoate (sodium acetate) and soda lime is taken in a hard glass test tube (as shown in Fig.) and heated over a bunsen burner.
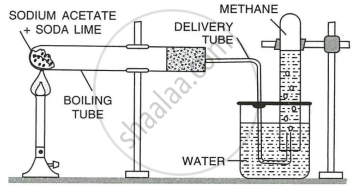
Reaction: \[\ce{CH3COONa + NaOH ->[CaO][300^\circ C] Na2CO3 + CH4 ^}\]
Collection: The gas evolved is collected by downward displacement of water since it is slightly soluble in water and is lighter than air.
shaalaa.com
Hydrocarbons: Alkanes
Is there an error in this question or solution?
Chapter 12: Organic Chemistry - EXERCISE-12B [Page 203]
