Advertisements
Advertisements
Question
Given below is the industrial process for the manufacture of ammonia gas.
Study the schematic diagram to answer the following questions.
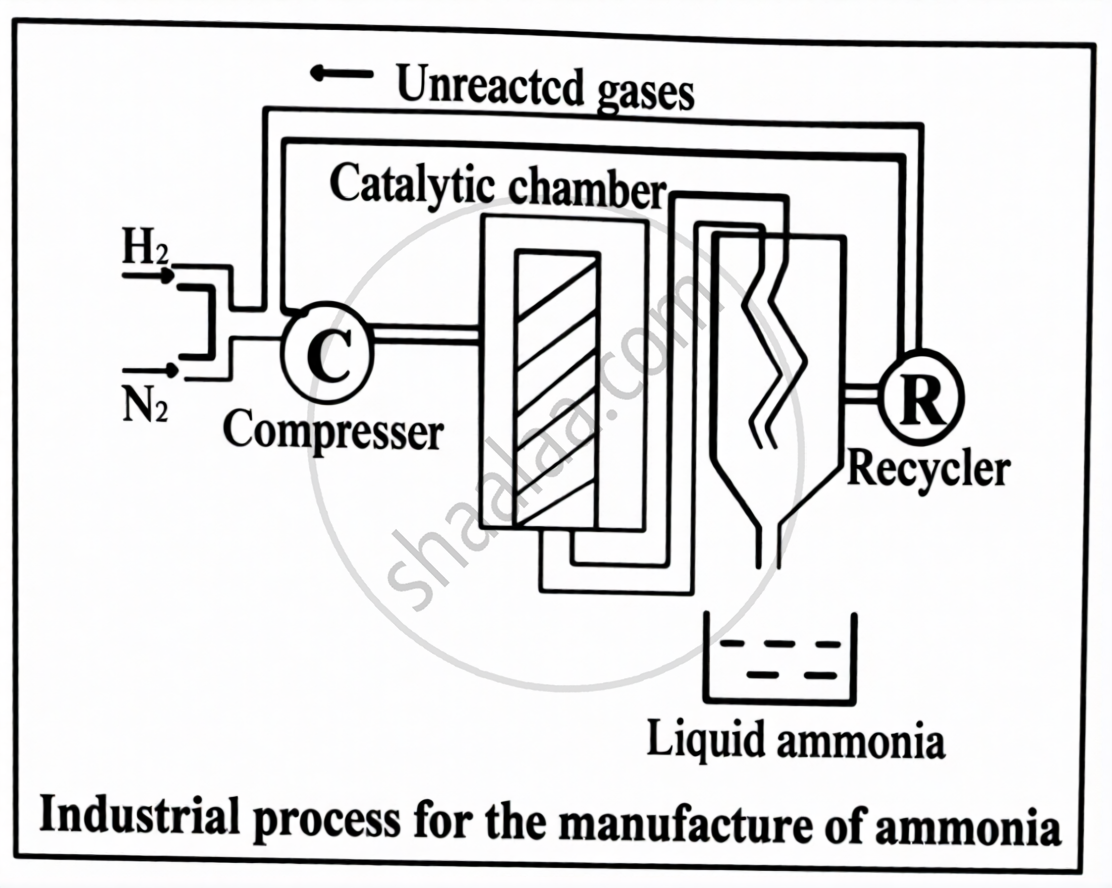
- Name the process.
- Which catalyst is used in the above process?
- In the above diagrammatic setup, how is ammonia gas separated from the unreacted gases to obtain liquid ammonia?
- Which two properties of ammonia gas can be demonstrated by the Fountain Experiment?
Very Long Answer
Advertisements
Solution
(a) The process is the Haber Process (also known as the Haber-Bosch process).
(b) The catalyst used is finely divided iron (industrial iron catalyst, often with small promoters such as molybdenum or Al2O3).
(c) Ammonia is separated through liquefaction.
In the cooling section (the zigzag pipes), the mixture of gases is cooled. Ammonia has a much higher boiling point than nitrogen and hydrogen, so it condenses into a liquid first and is collected, while the unreacted gases stay in a gaseous state and are recycled.
(d) The Fountain Experiment specifically demonstrates:
- High solubility in water: The gas dissolves so quickly that it creates a partial vacuum, drawing the water up.
- Basic (alkaline) nature: The change in the colour of the indicator (e.g., red litmus turning blue) proves that ammonia is a base.
shaalaa.com
Is there an error in this question or solution?
2025-2026 (March) Official Board Paper
