Advertisements
Advertisements
Question
Give balanced equations for the following reactions:
(1) Benzaldehyde reacts with hydrazine.
(2) Acetic acid reacts with phosphorous pentachloride.
(3) Acetone reacts with sodium bisulphite.
Answer in Brief
Advertisements
Solution
(1)
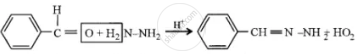
(2) \[\ce{PCI5 + CH3COOH-> CH3COCI + HCI +POCL3}\]
(3) \[\begin{array}{cc}
\ce{CH3\phantom{................} CH3\phantom{..} OH}\\
\phantom{}\backslash \phantom{....................}\backslash \phantom{..}/\\
\ce{C=O + NaHSO3<=>C}\\
\phantom{.}/\phantom{....................}/\phantom{..}|\phantom{...}\\
\phantom{.....}\ce{H3C}\phantom{................}\ce{H3C}\phantom{...}\ce{SO- 3Na+}
\end{array}\]
shaalaa.com
Concepts of Aldehydes, Ketones, and Carboxylic Acids - Chemical Properties of Benzaldehyde
Is there an error in this question or solution?
