Advertisements
Advertisements
Question
For the given close packed structure of a salt made of cation X and anion Y shown below (ions of only one face are shown for clarity), the packing fraction is approximately:
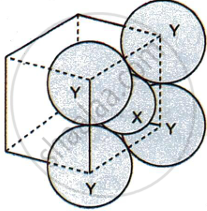
Options
0.74
0.63
0.52
0.48
MCQ
Advertisements
Solution
0.63
Explanation:
Assume the
X = r+
Y = r−
a = edge length of unit cell
2r− = a
`a/(r_−)` = 2
Also `r_+/r_-` = 0.414
Packing fraction = `(3 ("Volume of" r_+) + 1 ("Volume of" r_-))/"Volume of unit cell"`
= `(3 xx 4/3 xx pi r_+^3 + 1 xx 4/3 xx pi r_-^3)/(a/r)^3`
= `(4/3 pi [3((r_+)/(r_-))^3 + 1])/2^3`
= `(4/3 xx 3.14 [3 xx (0.414)^3 + 1])/8`
= `(4.176 xx [3 xx (0.070) + 1])/8`
= `(4.176 xx (0.21 + 1))/8`
= `(4.176 xx 1.21)/8`
= `5.05296/8`
= 0.63
shaalaa.com
Is there an error in this question or solution?
