Advertisements
Advertisements
Question
For the complex ion, [Fe(CN)6]3−:
Show the hybridisation diagrammatically.
Diagram
Advertisements
Solution
In [Fe(CN)6]3−, iron is in the +3 oxidation state, with the configuration [Ar] 3d5. The ligand CN− is a strong field ligand, which causes pairing of electrons in the 3d orbitals. This allows Fe3+ to use two 3d, one 4s, and three 4p orbitals for d2sp3 hybridisation.
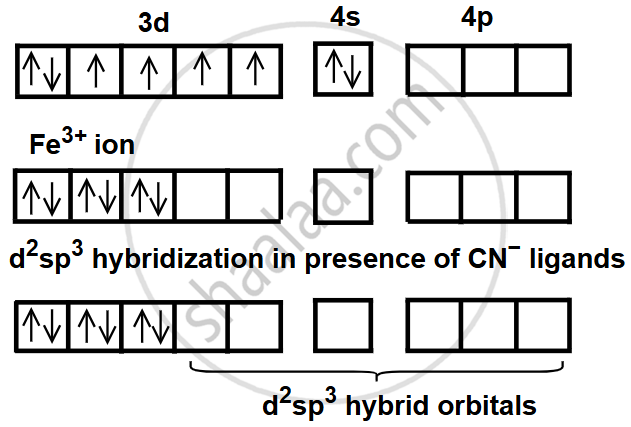
The hybridisation of the central metal ion in Fe(CN)6]3− is d2sp3.
shaalaa.com
Is there an error in this question or solution?
