Advertisements
Advertisements
Question
For the complex [Fe(en)2Cl2]Cl, identify: [5]
- the oxidation number of iron.
- the hybridisation and the shape of the complex.
- the magnetic behaviour of the complex.
- whether there is an optical isomer of the complex? If so draw its structure.
- IUPAC name of the complex. (At. no. of Fe = 26)
Advertisements
Solution
(a) Let x be the iron's oxidation number. Since it contributes zero charge, ethylenediamine (en) is neutral. One charge is contributed by each chloride ion (Cl). hence,
x + 0 + 2(–1) + (−1) = 0
x − 3 = 0
x = +3
∴ Oxidation number of iron, x = +3
(b) The two monodentate and two bidentate ligands make up the complex. Consequently, the shape will be octahedral, and the hybridization will be d2sp3.
(c) The oxidation state of iron is +3, resulting in the electron configuration of Fe3+: d5 given that the atomic number of Fe is 26. The presence of one unpaired electron in the d-orbital renders the complex paramagnetic.
![]()
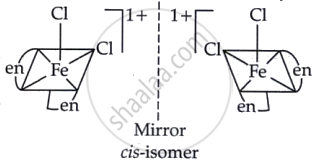
(d) The The cis-isomer of [Fe(en)2Cl2]Cl exhibits optical isomerism as it possesses non-superimposable mirror images resulting from the configuration of the chloride ions. The trans-isomer exhibits no optical isomerism due to its absence of chirality.
(e) Dichloridobis(ethane-1, 2-diamine)iron(II)chloride.
