Advertisements
Advertisements
Question
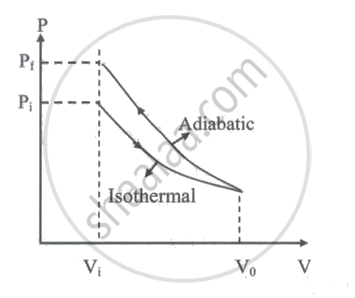
For an ideal gas with initial pressure and volume Pi and Vi respectively, a reversible isothermal expansion happens, when its volume becomes V0. Then it is compressed to its original volume Vi by a reversible adiabatic process. If the final pressure is Pf then which of the following statements is true?
Options
Pf = Pi
Pf > Pi
Pf < Pi
`"p"_f/"v"_0 = "p"_i/"v"_i`
MCQ
Advertisements
Solution
Pf > Pi
Explanation:
shaalaa.com
Is there an error in this question or solution?
