Advertisements
Advertisements
Question
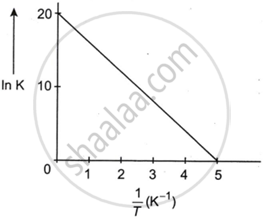
For a reaction, given below is the graph of ln K vs `1/T`. The activation energy for the reaction is equal to ______ Cal mol−1 (Nearest integer).
(Given R = 2 Cal K−1 mol−1).
Fill in the Blanks
Advertisements
Solution
For a reaction, given below is the graph of ln K vs `1/T`. The activation energy for the reaction is equal to 8 Cal mol−1.
Explanation:
We are given a graph of ln K vs `1/T`.
From the Arrhenius equation:
`ln K = ln A - E_a/R * 1/T`
It is of the form y = mx + c
Where slope m = `- E_a/R`
We need to find activation energy (Ea) in Cal mol−1, given R = 2 Cal K−1mol−1.
From the graph:
At `1/T` = 0, ln K = 2.
At `1/T` = 51, ln K = 0.
slope = `(0 - 20)/(5 - 0)`
= `(-20)/5`
= −4
i.e., `- E_a/R = -4`
But, Ea = 4 R
Ea = 4 × 2 = 8 Cal mol−1
shaalaa.com
Is there an error in this question or solution?
