Advertisements
Advertisements
Question
Find the total number of lone pairs of electrons in N2O3.
Numerical
Advertisements
Solution
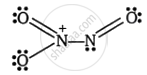
Three oxygen atoms and two nitrogen atoms make up this structure. One nitrogen atom makes a double bond with oxygen and a single bond with another nitrogen atom. Additionally, we can observe that one oxygen atom has a single bond and the other nitrogen has a double bond.
Now, let’s count the number of lone pairs present in this compound.
Total lone pairs = lone pairs on nitrogen atoms + lone pairs on oxygen atoms
Total lone pairs = 1 + 7 = 8
Therefore, we can conclude that the correct answer to this question is 8 lone pairs of electrons.
shaalaa.com
Is there an error in this question or solution?
